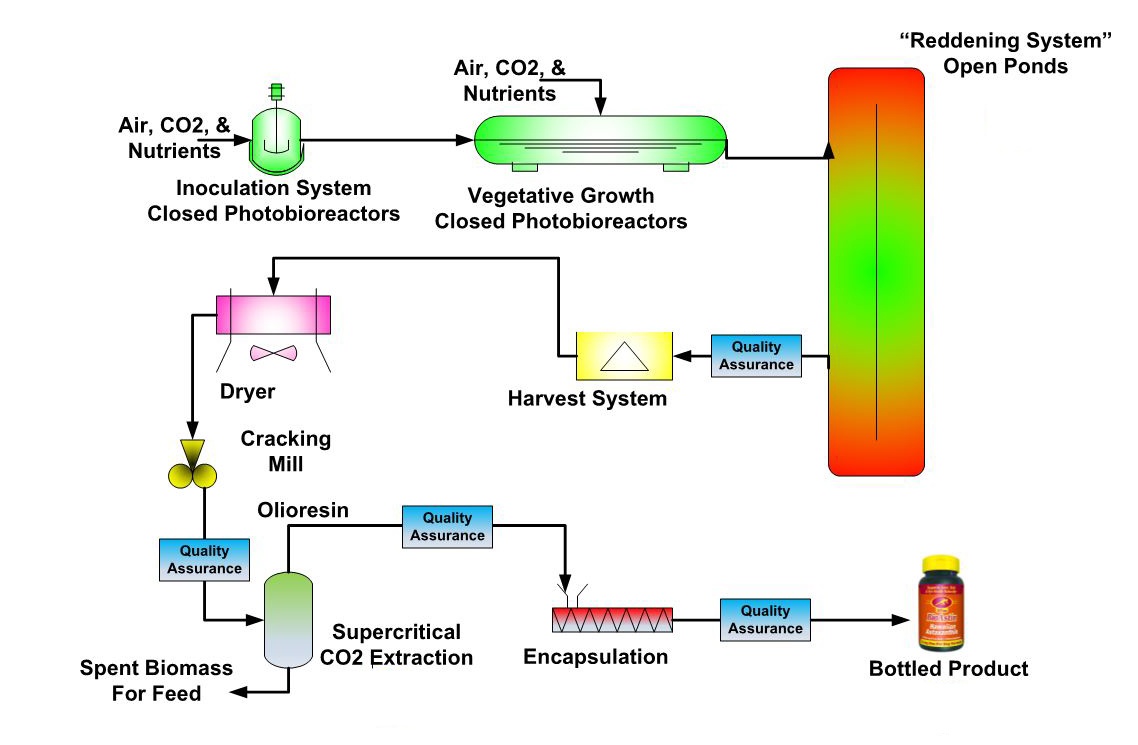
Astaxanthin Process

BioAstin Hawaiian Astaxanthin is produced using the microalgae Haematococcus pluvialis. Proprietary strains of Haematococcus are maintained in a biosecure laboratory and are used to inoculate 200 ml (one cup) shake flasks cultures—and the process begins.

Cultures of Haematococcus are scaled up in subsequent photobioreactors to 20 liters, then 400 liters, and on to 30,000 liters. The pH and temperature of the photobioreactors are carefully controlled and only filter sterilized air and water are allowed to enter to insure pure, dense, rapidly growing cultures of Haematococcus.

The final stage of the culture process is to subject Haematococcus to stress in open ponds under the intense Hawaiian sun. This stress causes Haematococcus to form a cyst and to rapidly start accumulating astaxanthin. An interesting fact is that Haematococcus cysts can be desiccated (dried) and remain dormant for decades and then spring back to life when conditions are good for growth. What makes this possible?---The antioxidant power of natural astaxanthin protects life essential ingredients such as DNA, RNA, and enzymes.

Quality Assurance runs through the BioAstin production process. The quality of the Haematococcus cultures is carefully monitored before harvesting as is the quality of the dried Haematococcus biomass and of the astaxanthin rich oleoresin after extraction (see #5). The final quality assurance testing is conducted on the finished softgel capsules and includes complete microbiological testing.

An astaxanthin rich oleoresin is extracted from the dried Haematococcus biomass using supercritical carbon dioxide. No toxic solvents such as hexane, heptane, acetone, or ethanol are used. Cyanotech is the only producer of natural astaxanthin that has fully integrated Haematococcus production with extraction of astaxanthin rich oleoresin allowing the Company to control and monitor all aspects of production. Cyanotech’s system is the world’s only 1,000 bar (14,700 psi) commercial scale supercritical extraction plant.

BioAstin Hawaiian Astaxanthin sofgels capsules are encapsulated to Cyanotech’s specifications by a third party manufacturer certified to operate under FDA-Good Manufacturing Practices (GMP) laws. Cyanotech also conducts regular inspection audits of the third party manufacturer. Every lot of BioAstin softgels undergoes full quality assurance testing, including complete microbiological testing, at Cyanotech prior to being released for sale.