Cyanotech Reports Financial Results for the Fourth Quarter and Fiscal Year 2021
KAILUA KONA, Hawaii (June 22, 2021) — Cyanotech Corporation (Nasdaq Capital Market: CYAN), a world leader in microalgae-based, high-value nutrition and health dietary supplement products, announced financial results for the fourth quarter and fiscal year 2021, ended March 31, 2021.
Commenting on the fiscal year results (changes shown vs. fiscal 2020), Cyanotech’s Chief Executive Officer, Gerald R. Cysewski, Ph.D., said:
“Net sales increased slightly over the prior year, however, gross profit was negatively impacted by higher spirulina costs per kilo driven by lower production volumes. We generated $2.4 million in cash from operations, deleveraged the balance sheet by $1.7 million, and increased our capital investments including the installation of a new tablet press.
“During the fourth quarter, we launched new labels for both our BioAstin® Hawaiian Astaxanthin® and Hawaiian Spirulina Pacifica® products. The new labels were well received in the marketplace.
“The coronavirus (“COVID-19”) pandemic has caused volatility in global markets. Our Company was declared an essential business in Hawaii and California, and we have remained open at full operations. We want to thank the team for their commitment to Cyanotech during a period of uncertainty.”
Fiscal Year 2021
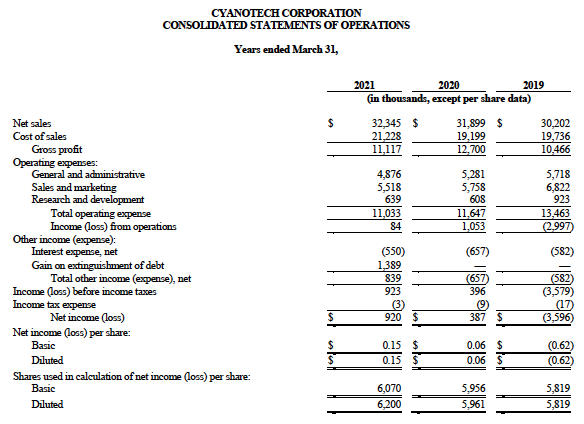
Cyanotech reported net sales of $32,345,000 for fiscal 2021 compared to $31,899,000 in fiscal 2020, an increase of 1.4%. Gross profit was $11,117,000, with gross profit margin of 34.4%, compared to gross profit of $12,700,000 and gross profit margin of 39.8%. Gross profit was negatively impacted by product mix and higher spirulina costs per kilo driven by lower production volumes in the current fiscal year. Operating income was $84,000 compared to operating income of $1,053,000.
Net income was $920,000 or $0.15 per diluted share, compared to net income of $387,000 or $0.06 per diluted share with the increase due to the forgiveness of the loan under the Paycheck Protection Program in the amount of $1,389,000, including accrued interest of $8,000.
Fourth Quarter Fiscal 2021
Cyanotech reported net sales of $9,438,000 for the fourth quarter of fiscal year 2021 compared to $8,634,000 in the fourth quarter of fiscal 2020, an increase of 9.3%. Gross profit was $2,771,000, with gross profit margin of 29.4%, compared to gross profit of $3,084,000 and gross profit margin of 35.7% in the fourth quarter of fiscal 2020. Operating loss was $24,000 compared to operating income of $228,000 in the fourth quarter of fiscal 2020. Net loss was $200,000, or ($0.03) per diluted share, compared to net income of $121,000, or $0.01 per diluted share in the fourth quarter of fiscal 2020.
Please review the Company’s Form 10-K for the period ended March 31, 2021 for more detailed information.
— Cyanotech will host a webcast at 8:00 PM EDT on Wednesday, June 23, 2021 to respond to questions about its operating results and other topics of interest. Interested parties are asked to submit questions to [email protected] before 12 p.m. EDT on Wednesday, June 23, 2021. The Company will respond only to relevant questions relating to the Company’s fourth quarter and fiscal 2021 financial performance and will not be accepting any questions or comments during the broadcast.
To join the webcast, please browse http://cyanotech.com/meet approximately five minutes prior to the start time.
About Cyanotech — Cyanotech Corporation, a world leader in microalgae technology for over 30 years, produces BioAstin® Hawaiian Astaxanthin® and Hawaiian Spirulina Pacifica®. These all natural, dietary ingredients and supplements leverage our experience and reputation for quality, building nutritional brands which promote health and well-being. The Company’s mission is to fulfill the promise of whole health through Hawaiian microalgae. Cyanotech’s BioAstin® offers superior antioxidant activity which supports skin, eye and joint health, as well as recovery from exercise*. Cyanotech’s Spirulina products offer nutrition that supports cardiovascular health and immunity.* All Cyanotech products are produced from microalgae grown at our 96-acre facility in Kona, Hawaii using patented and proprietary technology and are Generally Recognized as Safe (“GRAS”) for use in food products. Cyanotech sells its products direct to consumers at retail locations in the United States and online at www.nutrex-hawaii.com and also distributes to dietary supplement, nutraceutical and cosmeceutical manufacturers and marketers. The Company is regulated by the Food and Drug Administration. Visit www.cyanotech.com for more information.
*These statements have not been evaluated by the Food & Drug Administration. This product is not intended to diagnose, treat, cure or prevent any disease.
“Safe Harbor” Statement under the U.S. Private Securities Litigation Reform Act of 1995 Besides statements of present fact and historical fact, this press release may contain forward-looking statements. Forward-looking statements relate to the future and are subject to inherent uncertainties, risks and changes in circumstances that are difficult to predict. Our actual results may differ materially from those contemplated by forward-looking statements. We caution against relying on forward-looking statements. Important factors that could change actual, future results include: changes in sales levels to our largest customers, weather patterns in Hawaii, production problems, risks associated with new products, foreign exchange fluctuations, and availability of financing, as well as national and global political, economic, business, competitive, market and regulatory conditions. Other factors are more fully detailed in the Company’s annual Form 10-K filings with the Securities and Exchange Commission.
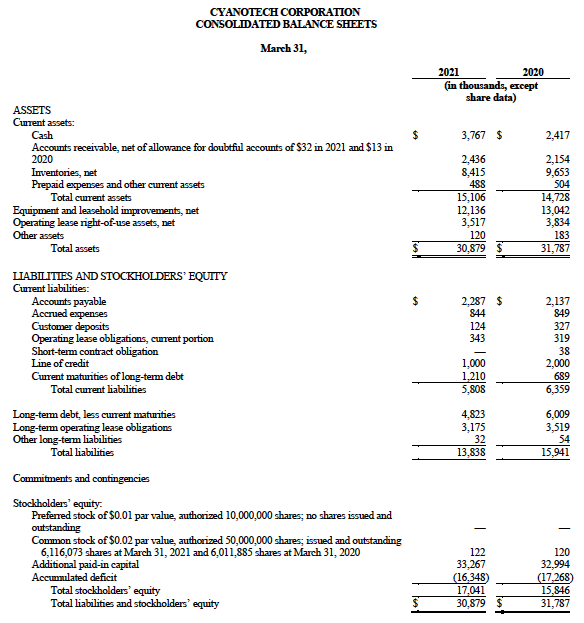
Financial Tables Follow: The following tables do not contain footnotes or other information contained in the Company’s Form 10-K for the fiscal year ended March 31, 2021, which can be found on the Cyanotech website (www.cyanotech.com) under Investors>Investor Filings upon filing. As such the following Financial Tables are provided only as a guide and other factors are more fully detailed in the Company’s annual Form 10-K filings with the Securities and Exchange Commission.